Epidermolysis bullosa (EB) is a rare group of genodermatoses affecting 1 in 17,000–50,000 live births worldwide (Siañez-Gonzalez et al, 2009; Maldonado Colín, 2016). It is characterised by mechanic fragility of the skin and mucous membranes, which causes blisters and erosions following minor trauma, as well as other systemic alterations resulting in several complications (Siañez-Gonzalez et al, 2009; Stevens et al, 2016).
Epidermolysis bullosa results from mutations in at least 18 different genes that encode proteins essential for maintaining the structural integrity of the skin (Maldonado-Colin et al, 2017). To date, more than 1,000 mutations have been identified that can arise de novo or be inherited through either an autosomal dominant or an autosomal recessive manner (Siañez-Gonzalez et al, 2009).
According to the latest consensus for EB reclassification in 2020, the four major EB types are as follows:
- EB simplex.
- Junctional EB.
- Dystrophic EB.
- Kindler EB (Has et al, 2020).
Each of these four types has its own particular phenotype, clinical manifestations and complications (Has et al, 2020; Pope et al, 2012). The main subtypes are classified by inheritance pattern, the recessive form being the most severe (Has et al, 2020).
Recessive dystrophic epidermolysis bullosa (RDEB) is caused by mutations in COL7A1, the gene encoding collagen VII, that forms the anchoring fibrils of the epidermal basement membrane (Siañez-Gonzalez et al, 2009; Kirkorian et al, 2014). The phenotype severe-generalised RDEB results from truncation mutations in the collagen VII gene, leading to absent or structurally deficient anchoring fibrils (Kirkorian et al, 2014). Clinically, this presents as extensive acute and chronic wounds; disability due to joint contractures; pseudosyndactyly; milia cyst formation; dystrophic or absent nails; and significant extracutaneous involvement including malnutrition, growth retardation, oesophageal strictures, osteoporosis, and an increased risk of developing squamous cell carcinoma, which can ultimately lead to death (Siañez-Gonzalez et al, 2009; Kirkorian et al, 2014).
Unfortunately, there is currently no cure for EB and the treatment is symptomatic and supportive, focusing on wound care and management of complications (Lara-Corrales et al, 2010; Pope et al, 2012). Dressings with lipido-colloid technology have been developed, demonstrating a reduction in healing time for lesions in EB patients (Letouze et al, 2004; Blanchet-Bardon and Bohbot, 2005).
The authors present the case of a paediatric patient with RDEB whose chronic lesions were treated with lipido-colloid technology dressings (UrgoTul and UrgoTul Ag/Silver), which led to an 80% improvement of the lesions within 6 weeks.
Case report
A 14-year-old male patient with a diagnosis of severe generalised RDEB presented with disseminated lesions affecting all body segments, predominantly the posterior trunk, buttocks, arms and legs with several months of evolution. The lesions consisted of multiple irregularly shaped ulcers of various sizes, along with numerous atrophic scars and multiple milia cysts on the earlobes. In addition, the patient had joint contractures, pseudosyndactyly of the four extremities, and total anonychia. His disease was congenital and genetic testing confirmed homocigocity.
Due to the chronicity of his lesions on the posterior trunk, topical management with a lipido-colloid dressing was initiated. The lesions were first cleaned with gauze impregnated with hypochlorous acid (Vashe wound solution), according to the manufacturer’s protocol, to reduce bacterial burden. Following this, a lipido-colloid dressing with silver (UrgoTul Ag/Silver) was applied (and changed every week), secured with a secondary dressing (gauze and bandage), for a 2-week duration. Subsequently, lipido-colloid dressings without silver (UrgoTul) were applied (changed every week) for 4 weeks due to the patient experiencing stinging and pruritus, which was attributed to the silver dressings. In addition to topical management, targeted antibiotic therapy (oral cephalexin) was administered for the first 10 days of treatment, based on an antibiogram of a wound culture taken due to the boy experiencing pain and malodor of the wound, which was positive for Enterococcus faecalis and Staphylococcus aureus. Since there was no improvement after 10 days of antibiotic treatment, the authors do not attribute the subsequent improvements reported to the use of antibiotics.
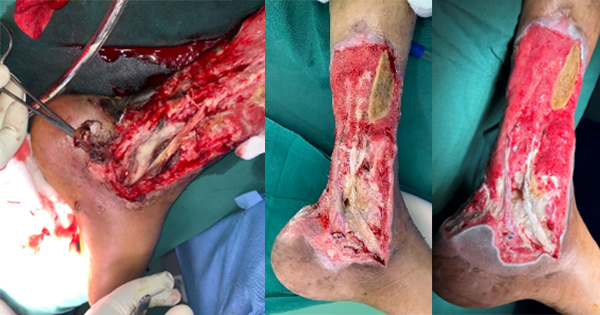
After 6 weeks of treatment, the patient showed a reduction of approximately 80% in lesion size [Figures 1 and 2], along with decreased pain and itching. The patient and caregiver reported that the dressing was easy to apply and remove, and that healing time was reduced compared with other dressings. During the first applications, stinging was reported during the first few minutes after applying the lipido-colloid dressing with silver (UrgoTul Ag/Silver) but resolved spontaneously. Other symptoms that presented were mild bleeding and temporary malodour. Upon switching to the lipido-colloid dressing without silver (UrgoTul), the patient reported decreased discomfort and better tolerance.
Discussion
Wound care is the cornerstone of management for EB, particularly in severe forms such as RDEB, where chronic wounds present significant challenges for patients and their caregivers (Pope et al, 2012; Stevens et al, 2016). A 2012 consensus approach to wound care in EB emphasised the importance of specialised, non-adherent dressings to reduce pain, prevent further skin trauma, and enhance wound healing (Pope et al, 2012).
Despite advances in wound care products, there is still no definitive cure for EB, and treatment remains primarily symptomatic and supportive (Kirkorian et al, 2014; Pope et al, 2012). Chronic wounds in RDEB patients often result from poor re-epithelialisation, increased bacterial load and mechanical skin fragility, leading to delayed healing and a higher risk of complications, such as infection and squamous cell carcinoma (Lara-Corrales et al, 2010; Pope et al, 2012).
Atraumatic dressing changes have shown to significantly improve quality of life for EB patients, as dressing removal is one of the most painful aspects of wound care (Blanchet-Bardon and Bohbot, 2005; Pope et al, 2012). Additionally, maintaining a moist wound environment, as achieved with lipido-colloid technology, has been shown to accelerate healing and reduce scarring, which is particularly relevant for patients with extensive chronic wounds (Blanchet-Bardon and Bohbot, 2005).
The lipido-colloid technology used in newer dressings (UrgoTul and UrgoTul Ag/Silver) represents an innovative approach in wound care, which aims to provide non-adherent, hydrating and antimicrobial benefits (Blanchet-Bardon and Bohbot, 2005) that are crucial in managing EB-associated wounds. These dressings are designed for the management of both chronic wounds, including venous and pressure ulcers, and acute wounds, such as second-degree burns, dermabrasions, traumatic injuries and postoperative wounds. The progressive release of silver ions in lipido-colloid dressings helps combat bacterial colonisation (Urgo Medical, 2025), which is a major impediment to wound healing in patients with chronic wounds. In contrast, non-silver dressings are more appropriate for wounds in granulation and healing phases, such as non-infected EB wounds, where a gentle non-adherent approach is essential to minimise trauma and support tissue regeneration.
Silver dressings provide broad-spectrum antimicrobial effects, making them particularly beneficial for critically colonised or infected wounds in EB patients (Pope et al, 2012). However, their use should be carefully considered, as silver may cause irritation, burning, or pain in the wound area, and requires a moist environment to be effective. Additionally, silver can affect keratinocyte production, which may impair wound healing if used unnecessarily; therefore, it should be reserved for wounds with a high bacterial burden (Pope et al, 2012; Leaper, 2012). In this case, our patient experienced transient stinging and mild bleeding upon application of the lipido-colloid dressing with silver, highlighting the importance of close monitoring and individualised wound care approaches. Upon transitioning to a lipido-colloid dressing without silver, these symptoms resolved spontaneously, and the patient reported better tolerance and reduced discomfort, reinforcing the need to balance antimicrobial benefits with potential adverse reactions in EB wound management.
We prescribed adjunctive treatment with targeted antibiotic therapy for one chronic wound that showed signs and symptoms of an acute infection; however, we consider that the observed clinical improvement resulted primarily from the effect of lipido-colloid dressings since the patient had previously experienced episodes of wound superinfection that were managed with culture-directed antibiotic therapy alone, which had not improved the healing of these chronic wounds.
Our findings align with previous studies investigating the efficacy of UrgoTul in EB patients. A previous study reported wound healing in an average of 8.7±8.5 days (range 1–36; Blanchet-Bardon and Bohbot, 2005). Additionally, 19 out of 20 patients achieved complete healing, except for one patient diagnosed with RDEB, whose wound was considered stagnant by the investigator despite a clear (50%) reduction in wound size (Blanchet-Bardon and Bohbot, 2005). This was similar to our patient, who showed significant, but not complete, wound reduction (80%). This suggests that wound healing outcomes in RDEB may be inherently limited by the nature of the disease, rather than the efficacy of the dressing itself. These findings highlight the ongoing need for advanced therapies to improve long-term wound healing outcomes in severe EB patients.
While innovative dressings like UrgoTul and UrgoTul Ag/Silver offer promising results, further research is needed to optimise wound management strategies and explore potential disease-modifying therapies, such as gene therapy and stem cell transplantation, which have shown some promise but remain experimental and high risk (Kirkorian et al, 2014). Until such therapies become widely available, wound management will remain the primary therapeutic approach for EB patients, focusing on reducing pain, controlling infection and maintaining skin integrity.
As limitations, we had a sample size limited to a single case exclusively of RDEB, and we did not evaluate the dressings in patients with other types of EB, which may present different responses to treatment. Additionally, the total treatment costs were not assessed, nor were they compared with those of other dressings.
Conclusion
Wound management and dressing changes are a part of daily life for patients with RDEB and remain a challenge for patients and their caregivers. Until a curative treatment is available, wound management will remain the main of treatment in patients with EB. In this context, lipido-colloid dressings represent a promising alternative to conventional dressings.
These studies emphasise the necessity of choosing the right dressing according to the specific attributes of the wound and the patient’s individual requirements, considering certain aspects like absorption efficiency, ease of application and pain minimisation during dressing replacement.